Oxygen(O), denoted as O on the periodic table, is a nonmetallic chemical element belonging to Group 16 (VIa), also known as the oxygen group. It is a colorless, odorless, and tasteless gas essential for the existence of living organisms.
Animals absorb oxygen, converting it to carbon dioxide, while plants utilize carbon dioxide and release oxygen back into the atmosphere. Oxygen readily forms compounds by reacting with almost any other element and participates in processes like combustion, often accompanied by the evolution of heat and light.
Table of Contents
History:
Oxygen was first discovered around 1772 by the Swedish chemist Carl Wilhelm Scheele, who obtained it through the heating of various substances. Independently, English chemist Joseph Priestley also discovered oxygen in 1774.
Later, French chemist Antoine-Laurent Lavoisier elucidated oxygen’s role in respiration and combustion, discarding the phlogiston theory.
Occurrence and Properties:
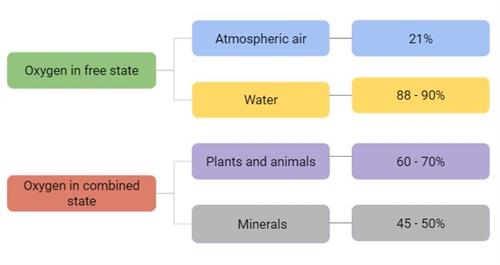
Oxygen constitutes 46 percent of Earth’s crust by mass. In the atmosphere, it makes up 21 percent by volume, while seawater contains oxygen at 89 percent by weight.
Naturally going on oxygen is a combination of solid isotopes, predominantly oxygen-16 (99.759 percent), oxygen-17 (0.037 percent), and oxygen-18 (0.204 percent). It has two allotropic forms: diatomic (O2) and triatomic (O3, ozone).
Allotropy:
Oxygen exhibits two allotropic forms: diatomic (O2) and triatomic (O3 or ozone). Ozone, with its three atoms not aligned along a straight line, is an important constituent in Earth’s upper atmosphere, protecting life from harmful radiation.
Preparative Methods:
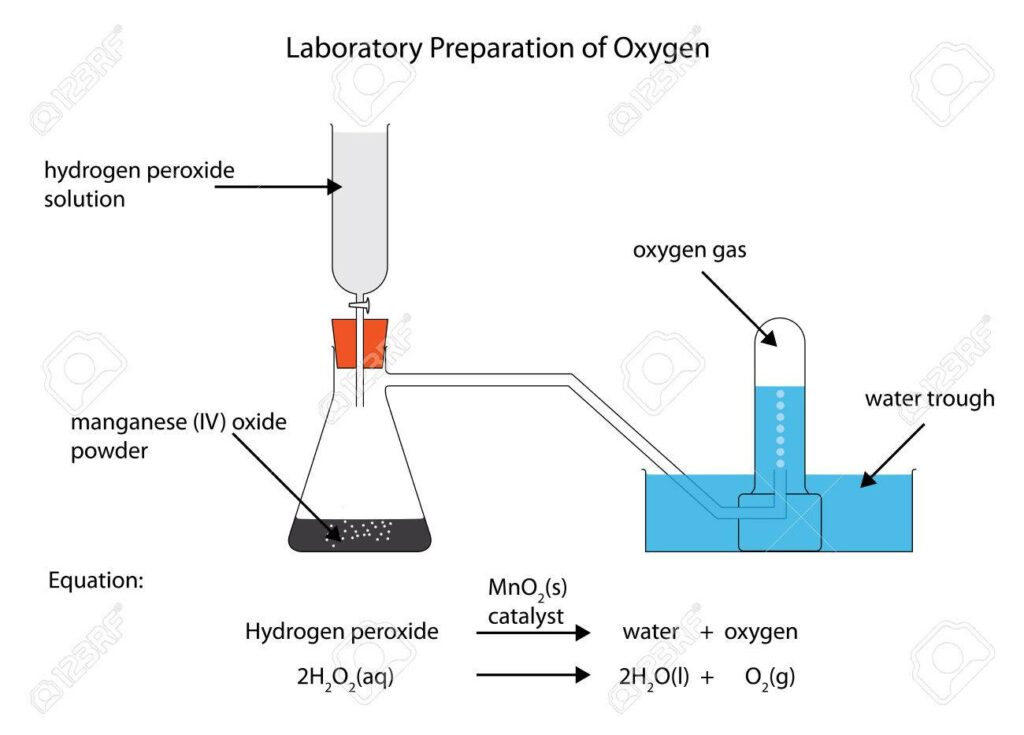
Various methods are employed to produce oxygen, including the thermal decomposition of salts like potassium chlorate, oxides of heavy metals, metal peroxides, or hydrogen peroxide. Electrolysis of water containing salts or acids is another method used in laboratories.
Commercial Production and Use:
In commercial applications, oxygen is often obtained by fractional distillation of liquid air. It is crucial in the steel industry for refining high carbon steel, wastewater treatment, waste incineration, and as rocket oxidizer fuel (LOX). Oxygen-enriched air is used in chemical manufacturing, medical applications, and diverse industrial processes.
Chemical Properties and Reactions:
Oxygen has high electronegativity and electron affinity, typical of nonmetals. It forms compounds through reduction, accepting electrons to create the oxide ion (O2-).
Oxygen reacts to form a variety of compounds, including oxides, peroxides, and organic compounds, demonstrating its significant role in various chemical processes.Oxygen is a vital element, central to life processes, industrial applications, and chemical reactions, shaping the world in numerous ways.
Also Read: hurrilet
FAQs
1. What is Õygen?
Õygen is a name that may be associated with various individuals, products, or services.
2. Is Õygen a specific brand or company?
Õygen could represent a brand or company, depending on the context in which it is used.
3. What does Õygen specialize in?
The specialization of Õygen can vary, and it is essential to consider the specific context in which the term is used.
4. Where can I find more information about Õygen?
To gather more information about Õygen, it’s recommended to search online or visit relevant websites or platforms associated with the term.
5. Can I contact someone related to Õygen for inquiries or support?
Contacting someone related to Õygen depends on the specific context and the availability of contact information associated with the term.
Conclusion
Õygen’s multifaceted presence suggests a diverse array of possibilities, be it in the realm of individual identities, products, or services. Its enigmatic nature invites exploration and research, prompting individuals to delve deeper into the context and significance of this distinctive term.
While the exact definition of Õygen may vary, its presence within various contexts serves as a catalyst for curiosity and discovery, encouraging individuals to uncover its true essence and meaning.
Read More:



+ There are no comments
Add yours